X Stop Spacer Reviews
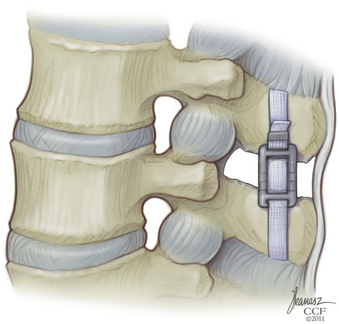
The X-STOP implant is placed between the. It is an all-titanium peek surrounded since end of 2004 device composed by an oval spacer one fixed wing one adjustable wing and one tissue expander Figures 1 and 2.

Interspinous Spacers A Wallis System B Diam System C X Stop And Download Scientific Diagram
X STOP is a titanium alloy device implanted between the spinous processes to relieve symptoms from low back spinal stenosis.

X stop spacer reviews. Ad Browse Our Huge Selection Of Discount Inhaler Spacer Products. Interspinous Process Spacers Video. The X-Stop interspinous distraction device has shown to be an attractive alternative to conventional surgical procedures in the treatment of symptomatic degenerative lumbar spinal stenosis 4 9It may be questioned however if the X-stop will be effective in patients with lumbar spinal stenosis caused by degenerative spondylolisthesis also. The X STOP Spacer for Lumbar Spinal Stenosis. Conflicting data exist concerning clinical outcome of the X-Stop interspinous spacer and little information is being published about implant survivorship and the need for revision surgery. The X-STOP System is a titanium implant with two components.
Your account has been temporarily locked. And a wing assembly consisting of an adjustable wing and locking screw. The surgical approach to the spine is from the back with the patient typically lying on his or her side or belly on the operating table. The X- Stop device received FDA premarket approval in 2005 with promising results in the short-term but further research demonstrated minimal benefit with longer-term follow-up along with relatively high complication rates. The researchers met the composite endpoint of the study demonstrating the Superion spacer was not inferior to the X-Stop spacer. Two-year results of interspinous spacer X-Stop implantation in 175 patients with neurologic intermittent claudication due to lumbar spinal stenosis.
LSS is the most common reason for back surgery in people aged 65 years and older in the United States. The X-STOP Spacer is the only interspinous spacer approved by the FDA in the United States to treat the symptoms of lumbar spinal stenosis LSS. To review the clinical outcome implant survivorship and reasons for failure after X-Stop implantation. The X-STOP which stands for Interspinous Process Decompression System is a titanium implant that is inserted into the back at the lumbar spine segment that has symptomatic spinal stenosis narrowing of the boney canal which can cause crowding of the nerve roots. X STOP is a nonfusion surgical implant for use in treating lumbar spinal stenosis. 1 An estimated 6 million Americans suffer from LSS with approximately 14 million being.
The X-STOP Spacer is the only interspinous spacer approved by the FDA in the United States to treat the symptoms of lumbar spinal stenosis LSS. X STOP received approval in 2005 from the United States Food and Drug Administration. Received the X-STOP spacer had signifi cantly better outcomes in each domain of the ZCQ specifi cally physical function symptom severity and patient satisfaction relative to CC patients11 Clinical adverse-event and quality-of-life data from the X-STOP spacer clinical trial were made available for this cost-effectiveness analysis. The Superion ISS is composed entirely of titanium alloy Ti6Al-4V ELI conforming to ASTM F136 The Superion ISS is intended to be implanted via minimally-invasive surgical methods using a set of proprietary accessory instruments provided by VertiFlex expressly for use with the Superion ISS device. The X-STOP procedure is performed under. LSS is the most common reason for back surgery in people aged 65 years and older in the United States.
X STOP is a titanium alloy device implanted between the spinous processes to relieve symptoms from low back spinal stenosis. X-STOP Interspinous Process Decompression IPD System Medtronics Minneapolis Minnesota. Insertion of the X-STOP spacer for spinal stenosis pain relief is a fairly straightforward surgical procedure and can be done in an operating room or specialty room at the hospital. Your account has been temporarily locked due to incorrect sign in attempts and will be automatically unlocked in 30 mins. The two current spacers that are FDA approved and commercially available are the Coflex and Superion devices. The X-STOP Spacer is the only interspinous spacer approved by the FDA in the United States to treat the symptoms of lumbar spinal stenosis LSS.
J Spinal Disord Tech. Summary of background data. The X-STOP Spacer is a small implant that can take the pressure off of pinched nerves due to lumbar spinal stenosis LSS. Interspinous process decompression with the X-STOP device for lumbar spinal stenosis. The X-STOP device was approved for use in the United States by the FDA in November 2005 and served as the control device in this trial. Lumbar spinal stenosis can cause pain and may limit physical function.
The goal is to review these two implants their indications and patient selection. 1 An estimated 6 million Americans suffer from LSS with approximately 14 million being. The X-STOP device has been described in detail elsewhere 24. 1 An estimated 6 million Americans suffer from LSS with approximately 14 million being. X-STOP Allows Machinist to Fulfi ll His Retirement DreamOne morning back in December 2004 when 58-year-old Mark Casale tried to get out of bed he fell to t. The result may be sustained pain relief.
At the time of database lock for PMA submission July 7 2014 946 183 Superion and 187 X-STOP IPD of patients enrolled in the study were available for analysis at the study completion 24-month post-operative visit. LSS is the most common reason for back surgery in people aged 65 years and older in the United States. The spinous processes are small stubby finger-like bones that slightly protrude off the back of each vertebral body. Kuchta J Sobottke R Eysel P Simons P. The X-Stop interspinous process decompression system is an interspinous spacer developed to treat patients with neurogenic intermittent claudication. Together the implants and manual instruments form a.
A spacer assembly consisting of a tissue expander an oval spacer and a fixed wing. A 4-year follow-up study. 99 percent of X-Stop patients. Interspinous spacers ISS have been developed as a less invasive strategy which may serve to avoid many of these risks. The Superion ISS cohort had a follow-up rate of 973 and the X. Leg pain was the predominant patient complaint.
Shop Save Now. The patients in both groups reported leg pain decreased in severity by 70 percent by two years after surgery. Back and leg pain from lumbar spinal stenosis LSS occurs when the space between the vertebrae is reduced causing bone or tissue to come in contact with the spinal nerve.

Lumbar Stenosis Back Pain Sciatica Advice By A World Renowned Spine Expert Chirogeek Com

Superion Interspinous Spacer In Situ A A P View B Lateral View Download Scientific Diagram
Interspinous Bumpers Clinical Gate
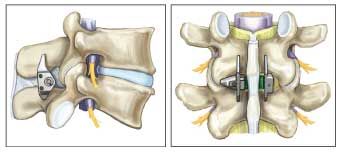
Managing Degenerative Lumbar Spinal Stenosis

Successful Implantation Of A Vertiflex Spacer At L2 3 Pacific Pain Physicians Interventional Pain Management Specialists
When To See A Surgeon For Spinal Stenosis
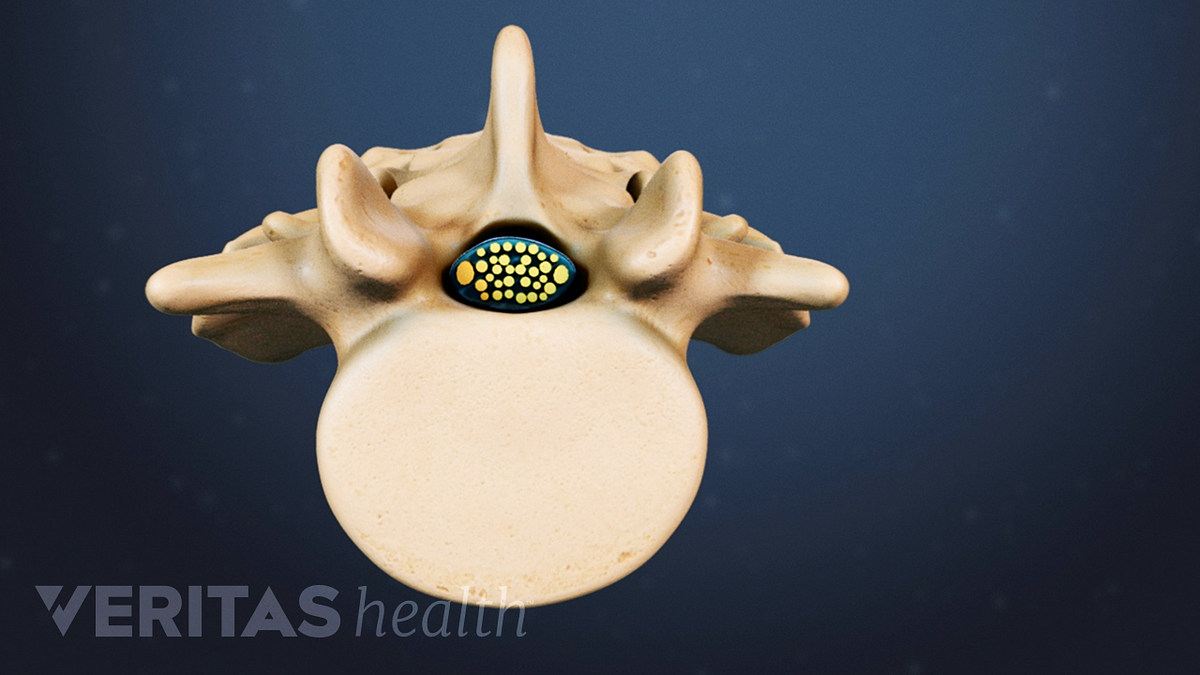
Spinal Stenosis Surgery The X Stop
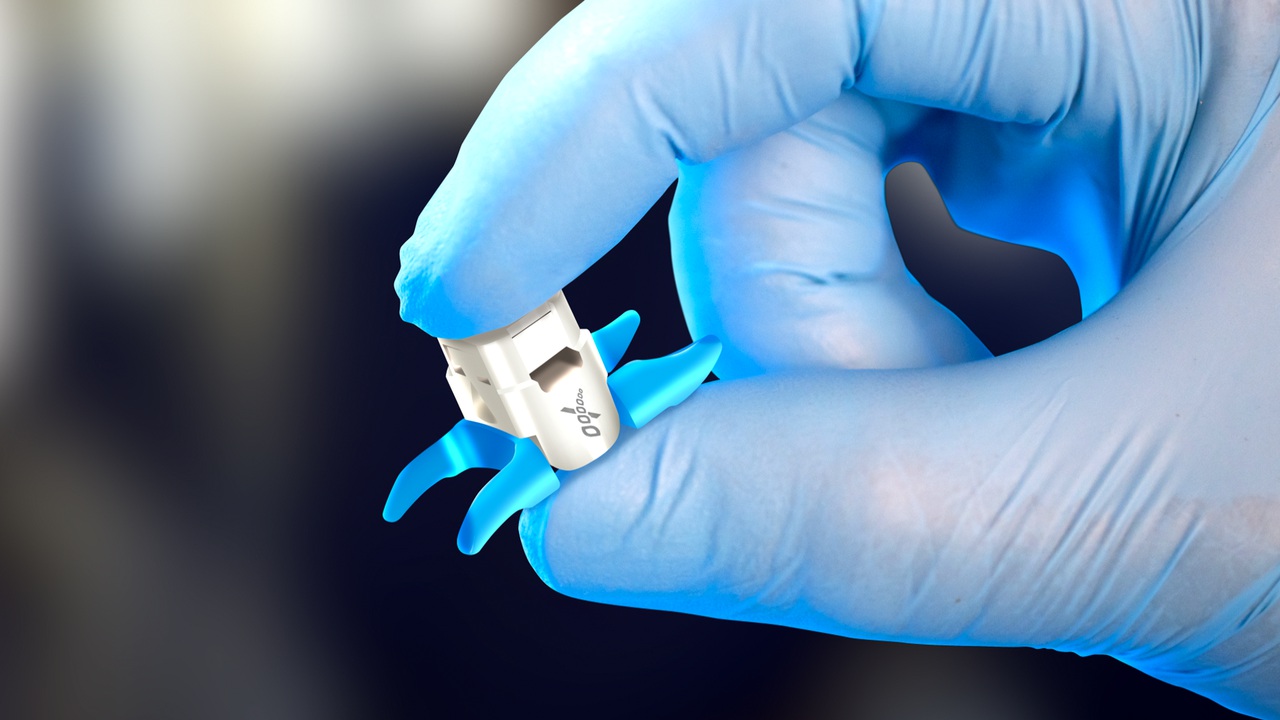
Vertiflex Superion Implant Cutting Edge Treatment Spinal Stenosis

Interspinous Spacer Relieves Painful Symptoms Of Lumbar Spinal Stenosis Study Shows

Interspinous Process Decompression X Stop Southern California Orthopedic Institute

Interspinous Spacer Relieves Painful Symptoms Of Lumbar Spinal Stenosis Study Shows

Coflex Device For X Stop 21 X Stop Ipd Procedure Is Indicated For Download Scientific Diagram

Posting Komentar untuk "X Stop Spacer Reviews"